|
This modification reduces andexanet alfa's procoagulant potential. 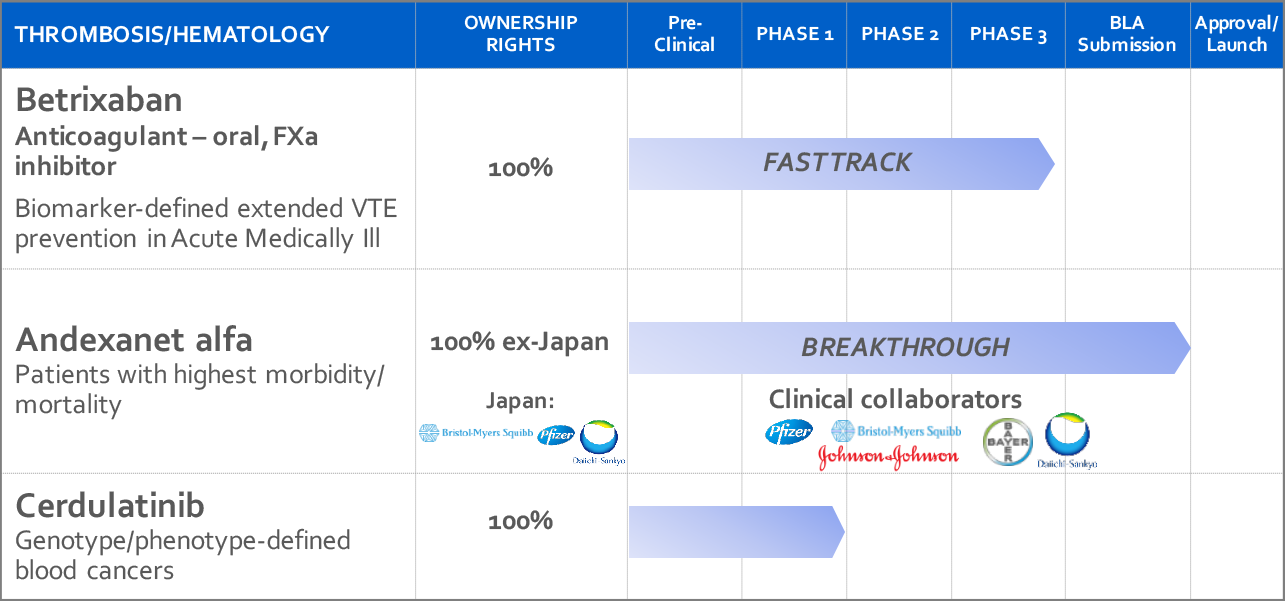
Andexanet alfa differs from native FXa due to the removal of a 34 residue fragment that contains the Gla domain. Pharmacology Mechanism of action Īndexanet alfa is a biologic agent, a recombinant modified version of human activated factor X (FXa). Īndexanet alfa has a boxed warning that it is associated with arterial and venous blood clots, ischemic events, cardiac arrest, and sudden deaths. Severe side effects may include blood clots or cardiac arrest. Adverse effects Ĭommon side effects include pneumonia and urinary tract infections. The FDA has demanded a randomised clinical trial: the first results are not expected before 2023. There was no relationship between hemostatic efficacy and reduced anti-Xa-activity. While people who were expected to die in 30 days were excluded from the study, 14% of participants died. This study reports results on 352 people and demonstrates a reduction of anti-Xa-activity while also showing an excellent or good hemostatic efficacy in 82%. The only published clinical trial is a prospective, open label, single group study. Studies in healthy volunteers show that the molecule binds factor Xa inhibitors and counters their anti-Xa-activity. There are no randomised clinical trials as of 2019. 
Medical uses Īndexanet alfa is used to stop life-threatening or uncontrollable bleeding in people who are taking rivaroxaban or apixaban. It was developed by Portola Pharmaceuticals. It was approved for medical use in the United States in May 2018. It works by binding to rivaroxaban and apixaban. Severe side effects may include blood clots, heart attacks, strokes, or cardiac arrest. Ĭommon side effects include pneumonia and urinary tract infections. 
It has not been found to be useful for other factor Xa inhibitors. Andexanet alfa, sold under the trade name Andexxa among others, is an antidote for the medications rivaroxaban and apixaban, when reversal of anticoagulation is needed due to uncontrolled bleeding.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
